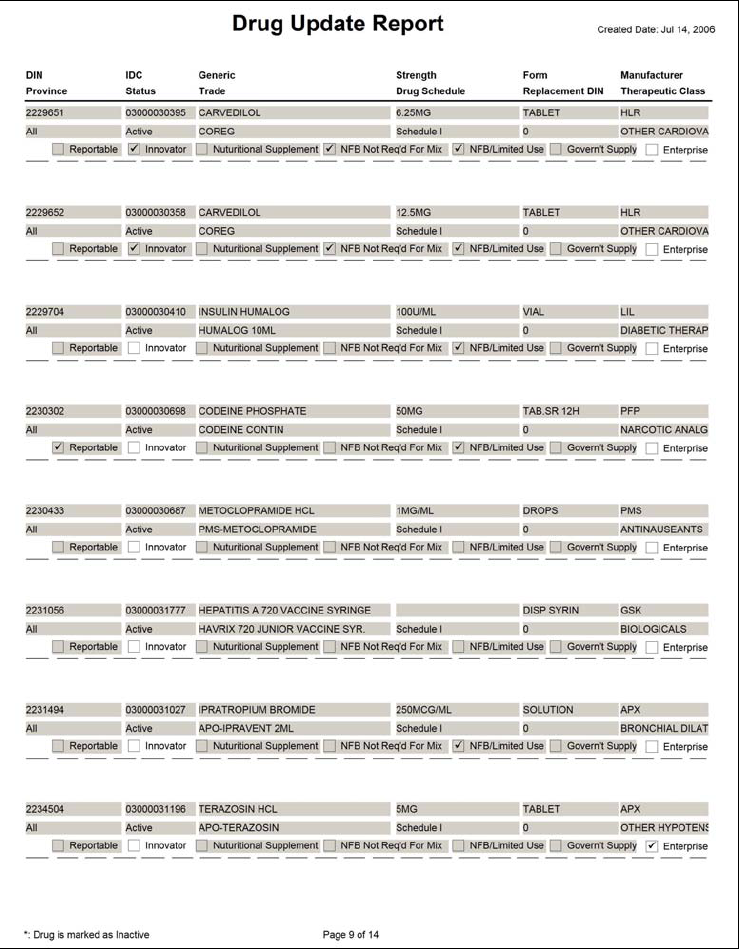
Drug Update Report
The Drug Update Report lists all drugs that have been added and updated since the last download. The report does not include updates that have the Action set to Do Later in the Review Overrides Drug Updates tab. The Drug Update Report does not include drugs that have received Supplier Price updates.
Note: Presently, the Drug Update Report only includes the updates to existing drugs. It does not include the new drugs that have been added. This will be corrected in a future version of PharmaClik Rx.
To view or print the drug update report:
- Press the More > Updates> Update Report. The Drug Update Report appears in a preview window.
- Use the scrollbars along the side and bottom of the window to view the report.
- To view the next page, press the Page Down key on the keyboard. To view the previous page, press the Page Up key on the keyboard.
Note: On the report, the fields highlighted in grey are fields that have been updated.
- Press the Print button to print the report.
- Press the OK button to close the window.
Sample Report
Use your mouse to rollover the fields below for more information.